Born Haber cycle/Calculation of lattice energy for sodium chloride/ Hess's law/ Thermodynamics - YouTube

How do you calculate the lattice energy of NaF using the Born-Haber cycle? (Delta Hsub for sodium is 107.5 kJ/mol) | Homework.Study.com

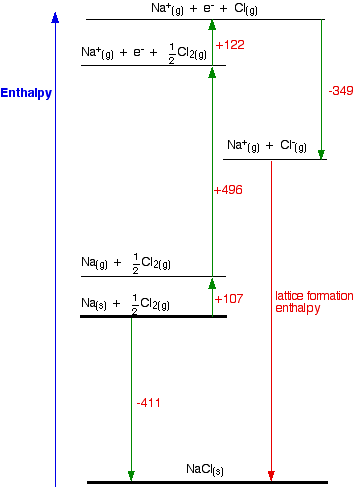
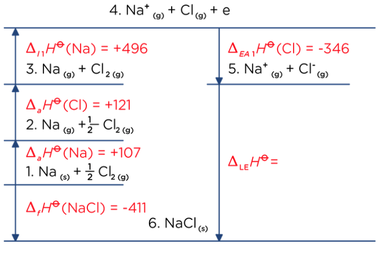
Calculate the lattice enthalpy of KCl from the following data by Born- Haber's Cycle. Enthalpy of sublimation of K = 89 kJ mol^(–1) Enthalpy of dissociation of Cl = 244 kJ mol^(–1)

Calculations using Born-Haber Cycles (5.1.4) | CIE A Level Chemistry Revision Notes 2022 | Save My Exams

Standard heat of formation of KI is - 78.31 kcal mol^-1 . Calculate its lattice energy from following information: IE (K) = 4.3 eV EA (I) = - 73.4 kcal mol^-1 Bond

Born Haber Cycle, Basic Introduction, Lattice Energy, Hess Law & Enthalpy of Formation - Chemistry - YouTube

Calculate the lattice enthalpy of LiF give that the enthalpy of :(i) Sublimation of lithium is 155.2 kJ mol ^-1 (ii) Dissociation of 1/2 mole of F2 is 75.3 kJ (iii) Ionization

SOLVED:Calculate the lattice energy of sodium oxide (Na2 O) from the following data: Ionization energy of Na(g)=495 kJ / mol Electron affinity of O(g) for 2 electrons =603 kJ / mol Energy